|
The Y group acts as a leaving group during the nucleophilic acyl substitution reaction.Electron withdrawing substituents increases the electrophilicity and makes the Carboxylic acid derivative more reactivethe substituent’s. The acyl group is the remainder of the molecule, which includes the carbonyl and the attached alkyl group. The reactivity of carboxylic acid derivatives is governed by this substituent as the electron-donating substituents reduce the electrophilicity and make the Carboxylic acid derivative less reactive.Carboxylic acid derivatives contain an electronegative heteroatom (typically oxygen, nitrogen, sulfur, or phosphorus) bonded directly to the carbonyl carbon represented by the symbol Y.Acyl phosphates are named following the IUPAC rules with the carboxylic acid ending with -oyl or -yl and -phosphate.Ģ1.2 Nucleophilic Acyl Substitution Reactions.Cyclic amides (lactams) use a Greek letter to identify the location of the nitrogen in relation the carbonyl group.Secondary or tertiary amides are named as primary amides are, but in additon an upper case N is used to designate the alkyl groups attached to the nitrogen atom, which are names as substituents.Primary amides are named following the IUPAC rules with the carboxylic acid ending with -oic acid being replaced by -amide.Thioesters are named following the IUPAC rules with the carboxylic acid ending with -oic acid being replaced by -thioate and the sulfide alkyl chain as a substituent.Esters are named following the IUPAC rules with the carboxylic acid ending with -oic acid being replaced by -ate and the alkoxy alkyl chain as a substituent.Anhydrides are named following the IUPAC rules with the carboxylic acid ending -acid being replaced by -anhydride.If the acyl groups are different, then they are named in. 
If both acyl groups are the same, then it is simply the name of the carboxylic acid with the word acid replaced with anhydride. 
Acid halide are named following the IUPAC rules with the carboxylic acid ending with -oyl or -yl and the halide ending in an -ide. A carboxylic acid anhydride ( RCOO OCR) is a carboxylic acid (COOH) that has an acyl group (RCO) attached to its oxygen instead of a hydrogen. 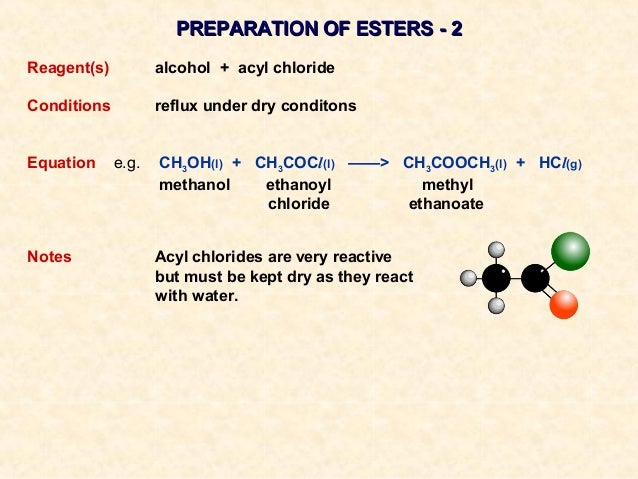
Nucleophilic acyl substitution reactions convert the carboxylic acid to one of the derivatives.Cyclic esters are referred to as lactones and cyclic amides are referred to as lactams.Many types of carboxylic acid derivatives, the chapter focused on acid halides (acyl halides), anhydrides, esters, amides, thioesters, and acylphosphates.Carboxylic acid derivatives are also known as acyl derivatives.Carboxylic acid derivatives formed when the hydroxyl group of the carboxylic acid is replaced by a different group.\)Ģ1.0 Chapter Objectives and Introduction to Carboxylic Acid Derivatives
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
